The ranges differ depending on what textbook you use, but I prefer categorizing differences of 0 - 0.5 as nonpolar covalent, 0.5 - 2.0 as polar covalent, and anything greater than 2.0 as ionic. The magnitude of that difference tells us what type of bonding is happening. Electronegativity again being a measure of how much an element attracts electrons where we can use the values found in the Pauling scale. You can differentiate the type of bonding occurring between atoms based on their electronegativity differences. So I'll address your second question first, and then move onto the first afterwards. It's just good to keep in mind the variability of classifying a bond's polarity. So using your professors numbers will get you by. Using simply electronegativity differences is most likely more than adequate for most general chemistry classes. So there are several valid ways to view bond polarity which can get increasingly complex depending on how detailed of an answer you'd like. So it's a wider view of bonding which includes the possibility of metallic bonding which electronegativity differences alone do not account for. It also always us to determine the percent ionic character of a bond too. The regions where bonds could possibly be form a triangle and this triangle is partitioned into ionic, polar covalent, nonpolar covalent, and metallic. The x-axis being the average electronegativity and the y-axis being the electronegativity difference. It's essentially a triangle which inputs electronegativity differences and averages from two bonding atoms and plots them in a 2D plane. Personally I prefer using something called the Van Arkel–Ketelaar triangle for determining bond type. 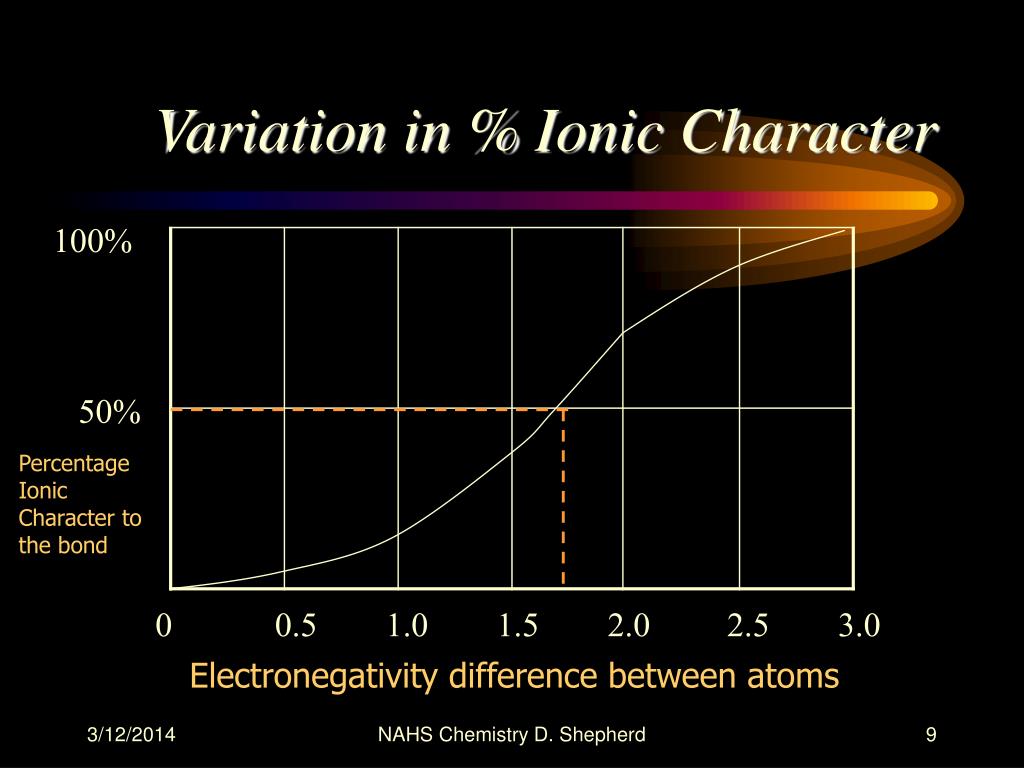
Bond dipole moments also correspond to electronegativity differences so they will agree with each other. A dipole moment is essentially a vector pointing towards the more electronegative atom showing where charge is accumulating in a bond. These ionic character percentages correspond to electronegativity differences (50% ionic character for example would means an electronegativity difference of 1.7) so they're alternative ways of gauging bond polarity, but percent ionic character reminds us though that bond type isn't black and white.Īnother way to quantify a bond's polarity is using its dipole moment. Predominantly covalent bonds would have an ionic character of <50%, and this could be further broken down into polar and nonpolar covalent. Linus Pauling, the chemist who developed the Pauling scale for electronegativity, used >50% ionic character as the mark for them a bond which was predominantly ionic. Pretty much no ionic bond reaches 100% ionic character though since there is at least some small degree of electron sharing. With the exception of completely nonpolar covalent bonds with a percent ionic character of 0%. Using percent ionic character, we can still classify bonds as being predominantly covalent or ionic but it reminds us that a bond is usually never completely one or the other. This lists bonds between 0% thru 100% with 0% being completely covalent and 100% being completely ionic. We can quantify these behaviors by something called percent ionic character. And ionic character can be seen as to what degree the electrons are transferred to one atom. 
In this sense covalent character can be seen as to what degree bonding electrons are shared and electron density exists between the atoms. However in reality bonds are certain percentages covalent and ionic. With this view bonds are either one or the other, but never both or a combination of the two. In that a certain electronegativity difference like 1.6 would be polar covalent, but slightly higher and it would be entirely ionic. The more conceptual second reason is that it views bonds in an absolute sense. The ranges you've listed look acceptable. There isn't a single set of ranges that are unanimously agreed on. There's a couple issues with this method though. Using electronegativity differences between atoms in a bond is a fair way to gauge a bond's polarity. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
